Uncovering the biology of cell rejuvenation to develop new therapies for age driven diseases
Cell aging is the largest driver of modern diseases
Aging mechanisms are common to the major modern diseases, and the source of aging is each and every somatic cell. Shift has built proprietary datasets that uncover the hidden biology of cell aging and rejuvenation, revealing single genes that rejuvenate multiple cell types more safely, small molecule targets and a connection to fibrosis.
This supports the development of new therapies for age-driven indications with potential to substantially increase lifespan, providing a path to $7 trillion of economic value.


Aging is reversed between generations
Each of us developed from a single cell passed down by our parents, yet we’re not born at our parent’s age and we begin our post-development lives in full health. The biology inherited from our parents is safely scrubbed, renewed and restarted, with evidence of a natural rejuvenation event in the embryo shortly after conception.
Shift finds a single gene that rejuvenates multiple cell types with safety
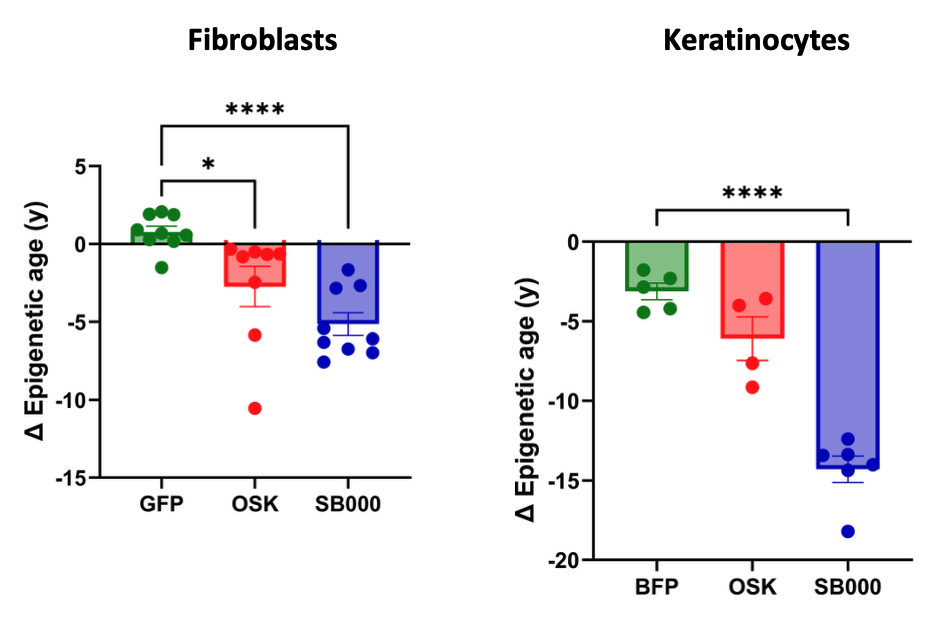
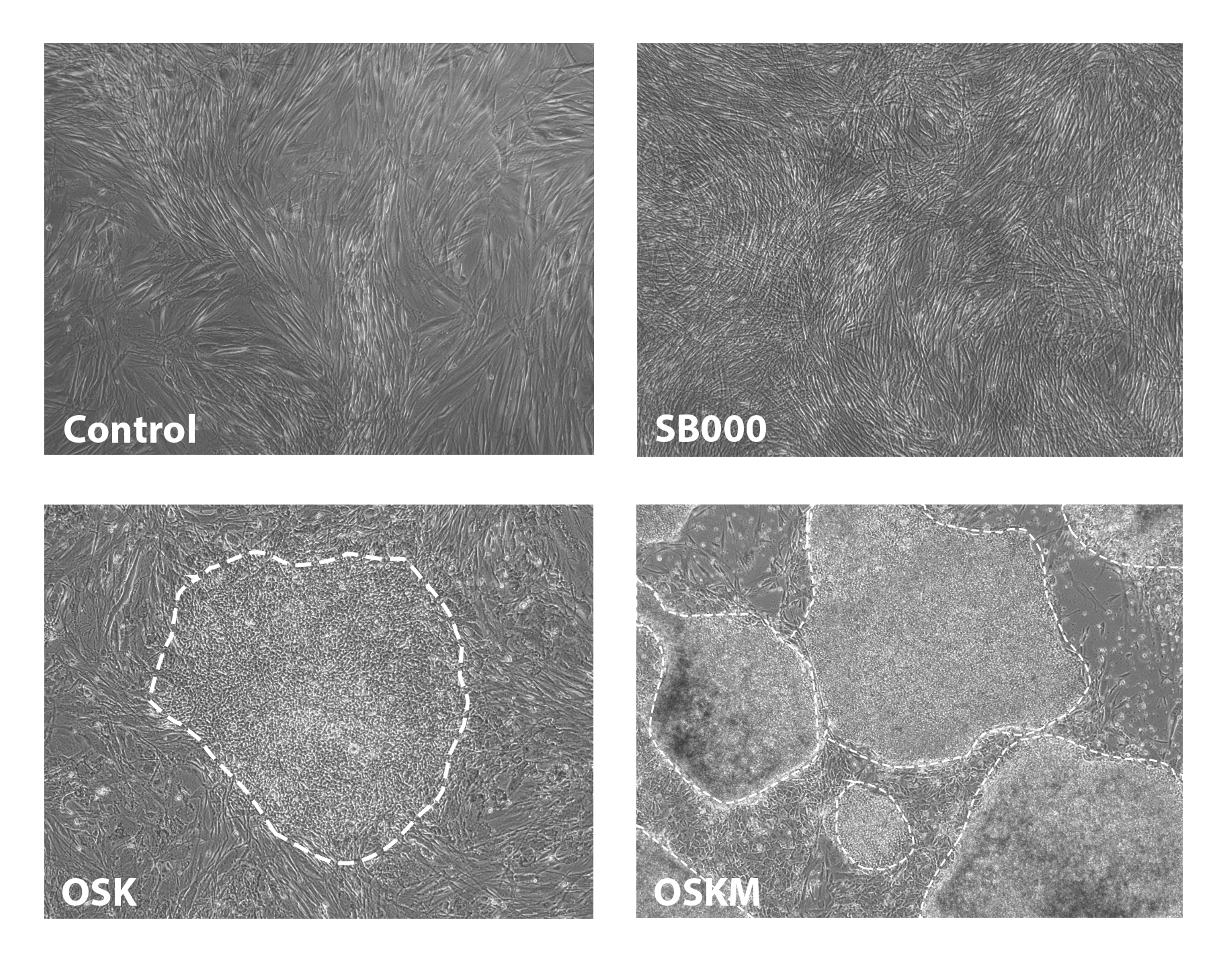
Yamanaka factors (OSKM) rejuvenate multiple cell types and extend the lifespan of mouse models, but were optimised to activate a tumor-inducing pathway, posing safety concerns for therapeutic development.
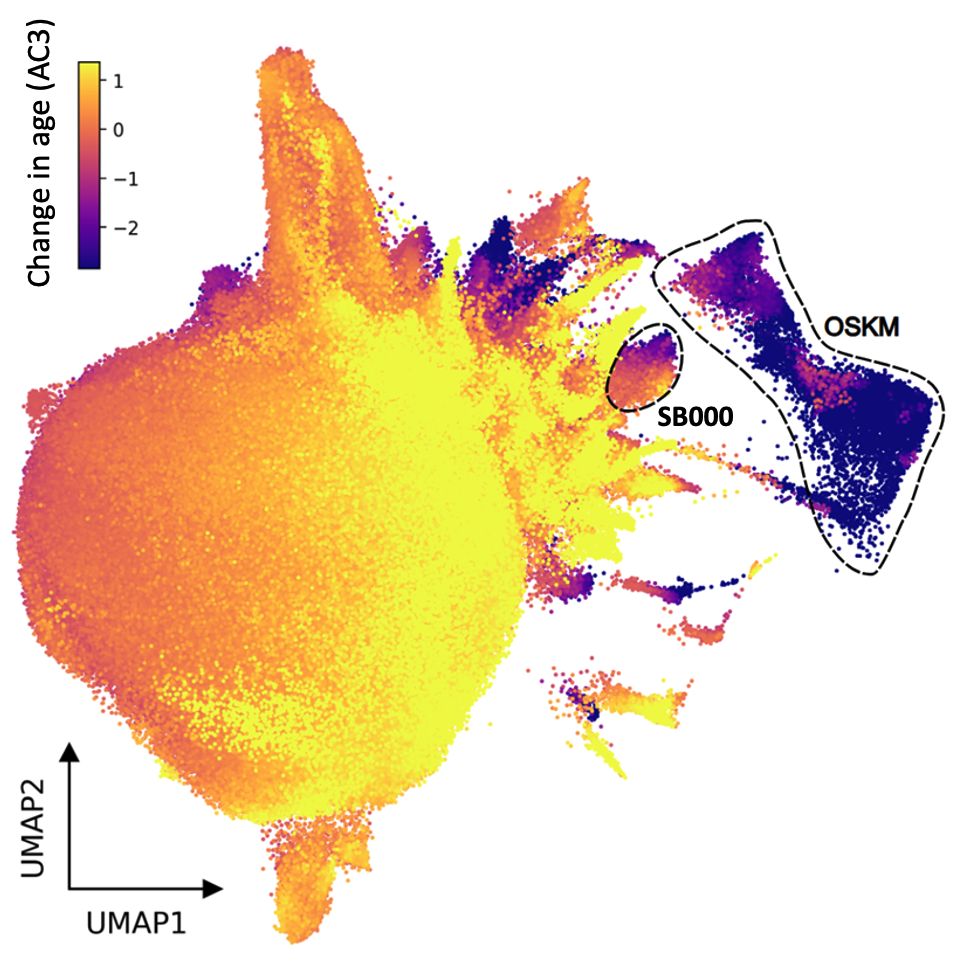
Shift's dataset enabled cell aging clock (AC3) and virtual cell have overcome this challenge, identifying genes that are individually sufficient to rejuvenate epigenetic age across multiple cell types (left or above) whilst decoupling the tumour inducing pluripotency pathway (below).
Leaders in virtual cells and cell aging clocks
Shift has assembled a world-class team of scientists that bridge machine learning and cell biology.

Senior advisor, Prof University of Toronto, Inventor of the cell simulator single-cell-GPT (scGPT)1

CSO and founder, PhD University of Cambridge, Inventor of the first accurate cell aging clock
Brendan received his PhD in Pharmacology from the University of Cambridge, where his focus was on basic research. First as an intern and then as a founder, Brendan began to prototype single-cell transcriptomic aging clocks, helping forge a new direction for Shift. Since 2021, Brendan has led Shift’s science team in the search for new rejuvenating interventions, with the belief that these discoveries could have a massive impact across healthcare.

Head of ML, MPhil University of Cambridge, Inventor of the most accurate aging clock2
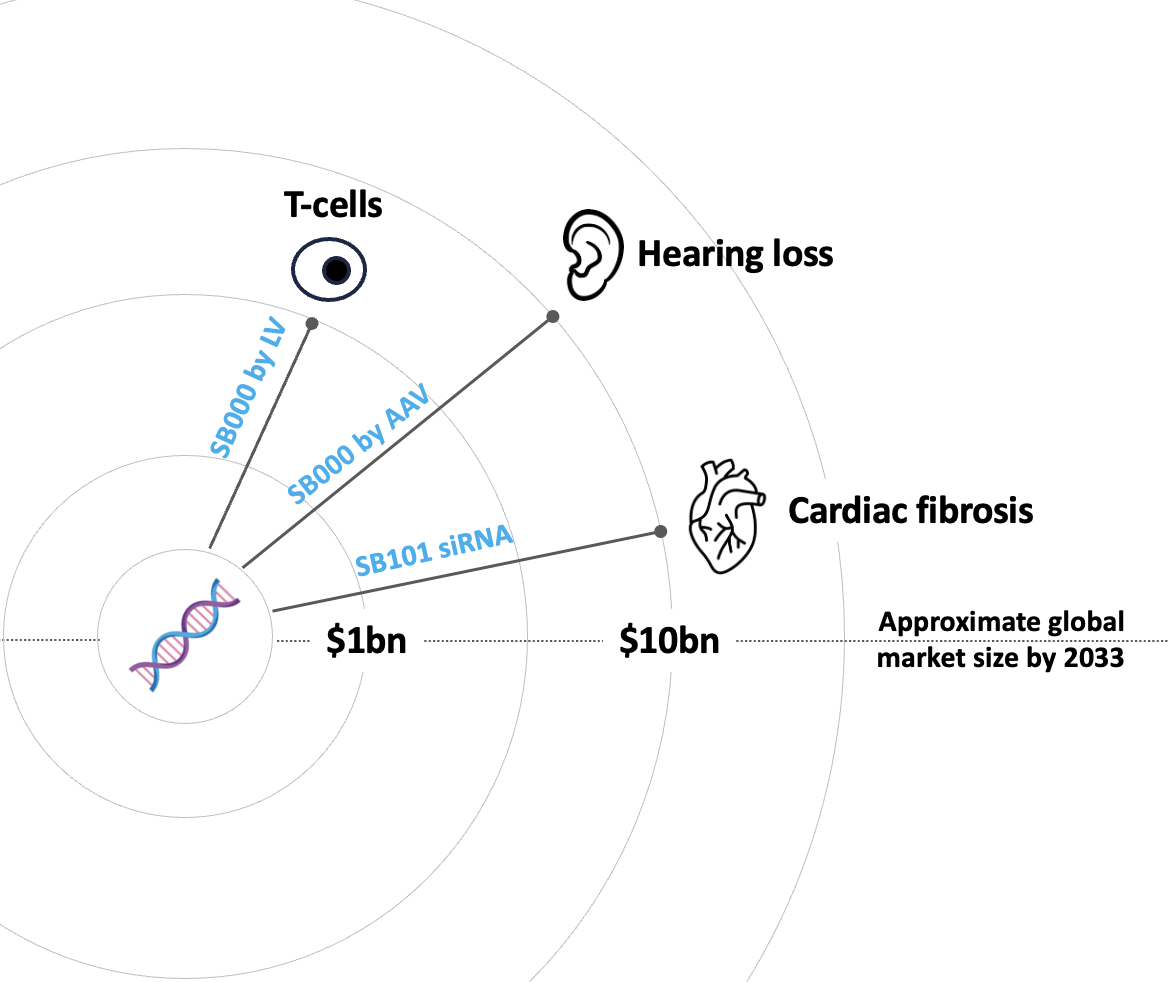
Shift is progressing a pipeline of cell rejuvenation assets
Shift's cell aging clock (AC3) and virtual cell reduce years of experiments to weeks, identifying 190 genes that individually affect the aging of real cells in vitro. Shift is progressing a pipeline of cell rejuvenation genes, siRNA and small molecules inhibitors of pro-aging genes across age-driven indications (see above).
Careers
Be first to find out about upcoming roles by following Shift's LinkedIn page

Leadership team
Current Investors
Latest news

Shift Bioscience proposes improved ranking system for virtual cell models to accelerate gene target discovery

Shift Bioscience identifies novel single-gene target for safer cellular rejuvenation therapeutics